Asphalt
Asphalt is produced through the refining of petroleum. It is a viscous adhesive that, along with aggregate, forms HMA pavement surfaces.
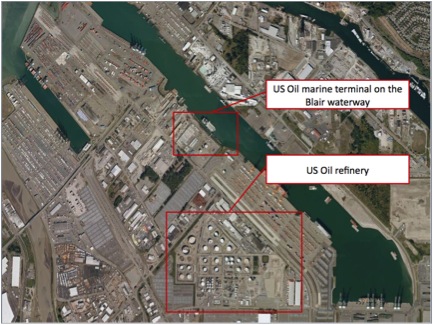
In this article we will follow the crude oil as it leaves the seagoing tanker at the Marine Terminal at US Oil and Refining Company in Tacoma, WA and becomes asphalt. Interestingly, much of the information about creating asphalt from crude oil comes from experimentation. The results of countless hours of experimentation will be laid as clearly as possible in this article.

Crude Oil
Crude oil is a flammable liquid found in nature. It is composed of hydrocarbons primarily made of carbon chains or rings, with hydrogen as the other primary constituent.
The amount of any particular hydrocarbon molecule can vary widely in a crude oil. This is apparent in the different classifications of crude oil. Light crudes can result in greater amounts of gasoline, kerosene, and naphtha. The heavy crudes will have more gas oil and residuum. As a general rule, the more carbon atoms in a hydrocarbon compound, the denser it is, and will have a higher boiling temperature. The opposite is true: fewer carbons means a lighter compound and lower boiling temperatures.
Crude oil may also be classified on the basis of sulfur content. Sweet crudes have 0.5% sulfur content or less and command a higher price than sour crudes, which have sulfur content of 1.5% or more. Sweet crude requires less processing than sour, hence the favorable price structure. The designation of sweet or sour may seem a bit unusual, but in the days when crude oil was sought after primarily as lamp oil, petroleum producers realized that high sulfur content crude oil would have an unpleasant smell when burned. People would taste the petroleum – if it had a high sulfur content, it would be sour; if not, it would taste sweet.
Transport
At this refinery, ships carrying crude oil arrive about every 10 days and off load about 400,000 barrels of crude which is pumped into large storage tanks. A substantial amount of the crude processed by US Oil is from the North Slope of Alaska, which is loaded at Valdez.
In the field, crude oil is pumped from the wells to a field separator, which is a wider section of pipe. As the crude flows into the wide pipe, the pressure inside the pipe drops. The lighter gasses (which may become natural gas at a separate gas processing plant) bubble out from the crude oil. Water separates at this point as well, and is drained from the pipeline at the field separator. Crude oil then continues on to transport.
Crude oil travels in many different ways to processing – truck, rail, pipe, or tanker. In the case of US Oil and Refining Co., tanker ship is the optimal form of transportation. US Oil and Refining Co. has an integrated marine terminal for accepting tanker ships.
The crude is pumped from the marine terminal at US Oil and Refining to the refinery through a dedicated 24” pipeline which is equipped with an emergency safety station and automatic crude sampler, which tests the crude oil for initial water content.
|
|
|
The Polar Adventure
The Polar Adventure is a double-hulled crude oil carrier, which conforms to the federal Oil Pollution Act of 1990. It was built in New Orleans, LA in 2004.
|
|
|
|
|
|
This ship is owned by Polar Tankers, Inc (a division of ConocoPhillips). It has a 125,000 ton deadweight, 895 ft. long by 152 ft. beam, and can carry more than 1 million barrels of crude oil.
Refining
Crude oil can be made of a thousand to several hundred thousand different hydrocarbon compounds. Each of them has different characteristics, such as weight, viscosity, and boiling point.
Crude oil will boil at approximately 150 degrees Fahrenheit, 450 degrees, 750 degrees, and 900 degrees. Each fraction vaporizes at these approximate temperatures, while others are left behind. When a container of crude is heated to a boil and held at one of the boiling temperatures, some of the crude will vaporize. This first boil is the lightest fraction (which may be butane or other light fractions.) The second boil will remove the gasoline, the third will remove naphtha and so on. This process of boiling and reboiling a batch of crude oil is the simplest form of refining, or distillation; it is commonly called batch distillation.
However, this process of boiling off fractions of crude oil is not the most efficient way to produce petroleum products. In the 1920s, fractional distillation was developed. In this method, petroleum is heated then piped into a distillation column or fractionation tower. Inside the tower or column are perforated trays, which catch liquid petroleum products at various levels and drain the separated components off to storage or further processing. The benefits to distilling in the towers include increased efficiency, less labor, and simpler facility construction. In fact, distilling crude oil is most efficient and least expensive when done in two steps: first, fractioning at atmospheric pressure, then feeding the residuum from the first column into a vacuum tower and distilling again.
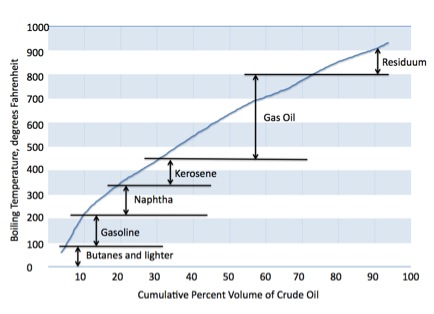
In boiling off separate fractions of the crude, a distillation curve can be plotted. This is a graph of temperature verses percent evaporated. Each type of crude oil will have a different, unique distillation curve that helps to describe it. As is shown in the following image, the distillation curve can be marked with the approximate locations of the fractions one will get out of the crude.
This process will be explored in more detail in the following sections.
Desalting
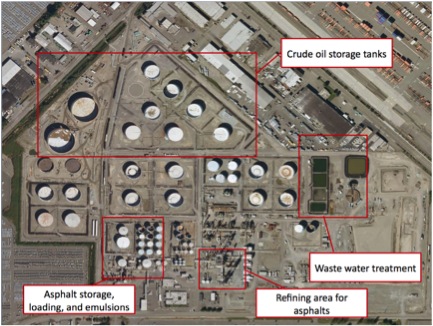
Once crude oil enters the refinery, water and salts are removed to make processing easier and less expensive. Sand, rust, minerals, salts, and other contaminants would foul a distilling column. Many of these will settle in the crude tanks and be drawn off as bottom sediment and water, but some will remain. Desalting also removes suspended solids in the crude. Suspended solids removal should be 60% or better in the desalting process – removal of particles greater than 0.8 micron should be near 80%.
If the salt content of a sample of crude oil is greater than ten pounds per 1000 barrels, desalting is necessary. Salt is dispersed throughout crude oil in tiny water droplets, an emulsion of water and salt in the oil. It will foul the distillation towers and corrode the metal works if not removed.
In order to take the salt out, fresh water is added to the crude oil to essentially wash the salt out. Chemicals to assist in breaking the emulsion may be added. The salt dissolves in the freshwater, and then the salty water drops to the bottom of the tank where it can be removed. This is carried out at about 200-300 degrees Fahrenheit. About 3-10% volume of water is added.
In especially stubborn cases, the crude oil may be passed through an electric field, where the water droplets can coalesce and settle out of the crude.

Distillation
The charge pump pumps crude from storage or the desalting tanks into the system. After desalting, crude oil passes through a series of heat exchangers to raise its temperature. This brings the crude up to about 550 degrees Fahrenheit through heat exchange with product and reflux streams.
A furnace further heats the oil up to about 700 degrees Fahrenheit and is sent to one of several fractionation towers or distillers. More than half of the crude oil is vaporized at this temperature. Inside the fractionation tower, which may have 30 to 50 trays, the heavier hydrocarbons (asphalt being one of these) settle on trays near the bottom, and the lighter ones (like butane) rise to the top as vapor or liquid and are removed.
The lighter fractions rise through perforations from tray to tray. The perforations are capped with a bubble cap, which forces vapor to move through liquid standing in a particular tray. In this process, heat transfers from vapor to liquid. The temperature of the vapor drops, and heavier compounds remaining in the vapor will then condense on each tray as the lighter fractions continue to move up the column.
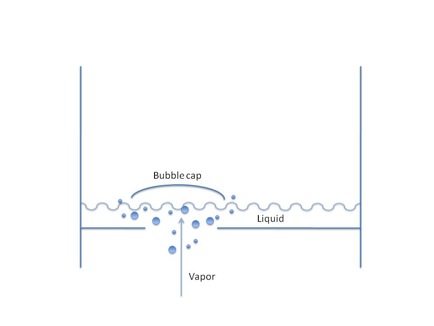
Fractions are removed as trays become full of liquid through a pipe called a side draw. Methane and ethane may be flared off. The heavier, tarlike hydrocarbons that settle at the bottom of a fractionation tower are called residuum.
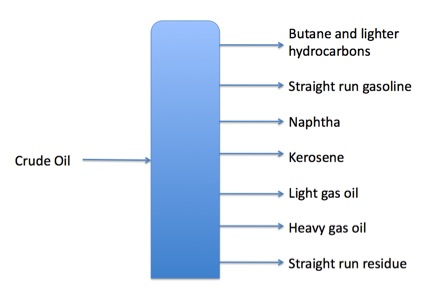
The range of temperatures at which the crude oil boils denote each fraction. This fraction is also called a cut. The key temperatures at which each product is separated is called a cut point. The lower temperature indicating a cut is the initial boiling point, or IBP. This is where the cut or fraction just begins to boil. The end point, or EP, is where that particular fraction has become completely vaporized.
Table 1: Some common cut points
| Under 90 degrees F | Butane and lighter |
|---|---|
| 90-220 degrees F | Gasoline |
| 220-315 degrees F | Naphtha |
| 315-450 degrees F | Kerosene |
| 450-800 degrees F | Gas oil |
| 800 degrees F and up | Residuum (including asphalt) |

This process can occur in one or several towers, and the towers may be at atmospheric pressure or can be pressurized, depending on the product and refinery.
Vacuum Flashing or Vacuum Distillation
Above 900 degrees Fahrenheit, cracking occurs. Cracking is when high temperatures cause the large hydrocarbon molecules to crack into smaller ones. This is undesirable as it is uncontrolled unless it happens in a catalytic cracking process. The heaviest cut points in a distilling column occur at about 750 degrees Fahrenheit.
Several fractions can continue to be separated if the boiling point of the mixture is lowered. This happens when the pressure is reduced. Therefore, it is more economical to distill at a reduced pressure to increase the number of products a refinery can produce. In vacuum flashing, the pressure is kept at 5% of atmospheric pressure or less. This combination of heat and low pressure is equivalent to distilling at a cut point of around 1000 degrees Fahrenheit.
Even though this process is occurring at high heat and low pressure, it is important to note that vacuum flashing is still a process of physical separation, and not chemical manipulation. No reactions happen in distillation or vacuum flashing. However, it is possible to break the larger hydrocarbons into smaller, lighter molecules – this is called cracking, as has been mentioned before.
The vacuum flashing assembly also contains a distributor, reflux, and vacuum pump. These assist in separating and capturing the final products. As the liquid and vapor enters the flasher, most of the liquid falls to the bottom of the column, while the rest of the liquid becomes droplets that rise with the vaporized part of the residuum. The distributor is meant to catch these droplets. A distributor is a mesh screen or tray of metal rings, and is several inches thick. These catch the droplets, which then coalesce and fall to the bottom.
Reflux is a process of guaranteeing that the heavier fractions do not get out of the top of the distilling column. In order to do this, some of the vapor is chilled and reintroduced to a lower tray. Whatever is still vapor at this point is sent off as one of the final products. Reflux in a vacuum flasher is usually an atomized spray at the upper portion of the column. The droplets of heavier product assist in the condensing of the vapor on one of the trays.
The vacuum pump keeps the pressure inside the vacuum flasher low. The pressure is typically kept at about 25 mm Hg to around 40 mm Hg, which is approximately 3 to 5% of atmospheric pressure. The vacuum pump also draws off vapor that hasn’t yet condensed, which is typically a mixture of water and some hydrocarbons.
To improve vaporization, the effective pressure is lowered further by the addition of steam into the tower. Effective pressure, which can be defined as the total absolute pressure minus the partial pressure of the steam, may be 10 mm Hg or less. A small decrease in pressure will lower the operation cost considerably. This is because the lower operating pressure significantly increases the volume of vapor per barrel vaporized, which means the vacuum distillation tower is a greater diameter than the atmospheric towers.

The streams drawn off of the upper part of the vacuum flasher are called flasher tops. This is comprised of light flashed distillate and heavy flashed distillate and are typically kept separate. However, both are typically sent to the catalytic cracker. The heavier part of flasher tops contains higher amounts of contaminants that will negatively affect the catalytic cracking process. Keeping the heavy and light flashed distillate separate allows for treating the heavy flashed distillate and encouraging more efficient use of the catalytic cracker later.
The last portion that is drawn off of the vacuum flasher is called the flasher bottoms. These liquids drain through a pipe which is quenched a bit to cool the fluid down. This prevents cracking that may still occur at high temperature. Flasher bottoms may become asphalt, or they may be sent on to be blended, may be cracked, or possibly sent to a coker.
The US Oil and Refining Vacuum Flasher
This distillation tower in the photos produces a bottom residual, which tests as a PG 64-22. It also produces flasher tops, or light vacuum gas oil, and flasher bottoms, or heavy vacuum gas oil, which are sent to a catalytic cracker offsite. From this tower, the asphalt is moved to storage tanks. The feedstock for this tower has been processed removing components such as naphtha and diesel.


Fluid Catalytic Cracking
If the refinery is equipped for it, longer hydrocarbons can be broken into shorter ones with fluid catalytic cracking, or “cat cracking.” This is done at high temperatures and pressures, with the assistance of a catalyst, a chemical which helps a reaction along without being used up in the reaction itself. US Oil and Refining Co. contracts with another facility to perform these operations.
First, the fractions may need to be treated to remove impurities. Then flasher tops and bottoms are mixed with a catalyst in the cat cracker to create smaller and more valuable molecules. This is done at 650 – 1100 degrees Fahrenheit. Cat cracking breaks molecules, but will form the full range of hydrocarbons as well as coke, or pure carbon. These can then be distilled as in regular operations, to create a wider range of hydrocarbons from the heavies coming from straight run distillation.
Finally, the different fractions are combined to create the final product. Other additives may be mixed in with the fractions at this time. This may produce gasoline of different octane ratings, or asphalt at different PG ratings.
Asphalt Storage and Emulsion
|
|
|
|
|
|
Some flasher bottoms, such as those at the US Oil and Refining Co. can be used immediately as asphalt. In this case, the residuum from the vacuum flasher tests as a PG 64-22, but this is not true for all refineries. The residuum in this case is a black or dark brown, nearly solid material.
Several grades of asphalt can be created at a refinery by creating blocked out runs, which means the vacuum flasher is run at one setting to produce a very hard asphalt. Then the settings can be changed to produce a soft asphalt, these can be blended to produces a wide variety of intermediate asphalts. Other additives may be included, as well.
Different additives may be added to asphalt depending on mix specifications. Some of the most common additives are EVA, a polymer which makes the mix more workable, especially in colder conditions, Sasobit, a wax which allows the asphalt to be mixed at lower temperatures, and SBS, a thermoplastic rubber which offers tensile strength and strain recovery.
|
|
|
US Oil and Refining Company Production
The refinery currently processes about 38,000 barrels of crude oil per day, and is open 24 hours per day, 365 days per year. Over the span of a year, US Oil produces:
- About 3 different asphalt emulsions
- About 15 different PG grades
- The most commonly specified PG grade at US Oil is PG 64-22.
Gasoline typically has 10% ethanol by volume added to all blends—which in turn is sold to companies such as Exxon, Costco, etc. The formulation for gasoline is changed seasonally, 6 times per year.
Asphalt is composed of the heavier, longer hydrocarbon chains from crude oil. At US Oil and Refining Company, the most common grade of PG binder, PG 64-22, comes straight from the vacuum tower. Other PG binders can be made with simple alterations to the formula as previously noted.
Fuel Products at US Oil & Refining Co.
- Fuel products
- All grades of unleaded gasoline and E10.
- Ultra low sulfur diesel fuel
- Jet fuel (JP-8 and Jet A)
- Asphalt products
- Cutback MC-Cold Mix
- Emulsions
- CMS-2
- CRS-2
- CRS-2P
- CSS-1
- CSS-1H
- STE-1
- Paving
- AC 2.5, 5 or 7
- Arctic AC-5
- AR 2000
- PBA-2, 5 or 6
- PG 52-28
- PG 58-22, 28, 34, 40
- PG 64-10, 22, 28
- PG 70-22
- PG 76-22
Refinery Laboratory
US Oil and Refining Company performs a multitude of tests to insure each of its products meets specifications for each product and its use. In order to do this, they run a large laboratory for testing asphalt, fuel oil, and gasoline. The remainder of this section will primarily focus on asphalt tests and specifications.

Specification Sheets
Asphalt is defined by its specifications. These criteria predict how the asphalt will react in the field, in a variety of temperature ranges. The most popular specification system is the Superpave performance grading (PG) binder system. Superpave performance grading is based on the idea that an HMA asphalt binder’s properties should be related to the conditions under which it is used. For asphalt binders, this involves expected climatic conditions as well as aging considerations. Therefore, the PG system uses a common battery of tests (as the older penetration and viscosity grading systems do) but specifies that a particular asphalt binder must pass these tests at specific temperatures that are dependent upon the specific climatic conditions in the area of use.
Dynamic Shear Rheometer

The dynamic shear rheometer (DSR) is used to characterize the viscous and elastic behavior of asphalt binders at medium to high temperatures. This characterization is used in the Superpave PG asphalt binder specification. As with other Superpave binder tests, the actual temperatures anticipated in the area where the asphalt binder will be placed determine the test temperatures used.
Note that in the specification sheets, the dynamic shear measurement is noted for PG 64-22 as:
- @64°C = 1.00 kPa (min)
- After RTFO
- @64°C = 2.20 kPa (min)
- After PAV 100°C
- @25°C = 5000 kPa (max)
Kinematic Viscosity

The kinematic viscosity of a liquid is the absolute (or dynamic) viscosity divided by the density of the liquid at the temperature of measurement. The 135° C (275° F) measurement temperature was chosen to simulate the mixing and laydown temperatures typically encountered in HMA pavement construction.
Viscosity requirement for PG 64-22:
- Viscosity @ 135°C = 3.0 Pa-s (max)
- Test is run on straight run PG (no aging)
Flash Point of PG 64-22

A typical flash point test involves heating a small sample of asphalt binder in a test cup. The temperature of the sample is increased and at specified intervals a test flame is passed across the cup. The flash point is the lowest liquid temperature at which application of the test flame causes the vapors of the sample to ignite. The test can be continued up to the fire point – the point at which the test flame causes the sample to ignite and remain burning for at least 5 seconds.
Creep Stiffness for PG 64-22

The Bending Beam Rheometer (BBR) test provides a measure of low temperature stiffness and relaxation properties of asphalt binders. These parameters give an indication of an asphalt binder’s ability to resist low temperature cracking. The BBR is used in combination with the DTT to determine an asphalt binder’s low temperature PG grade. As with other Superpave binder tests, the actual temperatures anticipated in the area where the asphalt binder will be placed determine the test temperatures used.
Creep stiffness is measured with the BBR:
- @-12°C, S = 300 MPa (max)
- @-12°C, M-value = 0.300 (min)
Pressure Aging Vessel
|
|
|
The pressure aging vessel (PAV) provides simulated long term aged asphalt binder for physical property testing. Asphalt binder is exposed to heat and pressure to simulate in-service aging over a 7 to 10 year period.






















